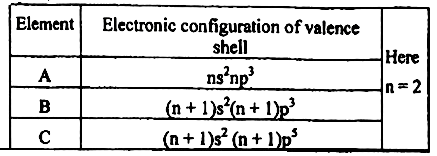Chemistry 1st Paper CQ-Cumilla Board-2022
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
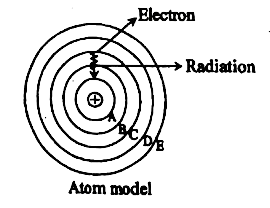
1. At temperature and , pressure the below reaction is at equilibrium state-
Here and are the elements of 3 rd period and is dissociated .
CB 22
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
4. A student wrote the following two electronic configurations for and :
(according to Aufbau rule)
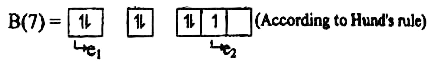
CB 22
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো