Chemistry 2nd Paper CQ-Chittagong Board-2017
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
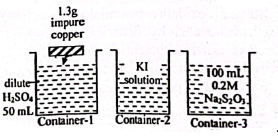
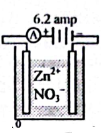
1. (i) Natural gas + Air
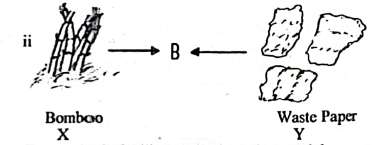
is an organic fertilizer and is study materials.
Chittagong Board-2017
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
2. 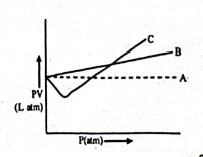 is produced by thermal decomposition of :
is produced by thermal decomposition of :
Chittagong Board-2017
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো