Chemistry 2nd Paper CQ-Barishal Board-2017
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
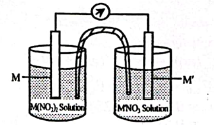
1. (i)
(ii)
[B compound is a position a isomer of compound
Barishal Board 2017
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
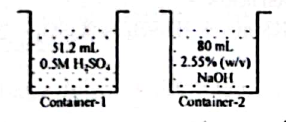
3. 
' ' is an acidic hydrocarbon containing two carbons.
Barishal Board 2017
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
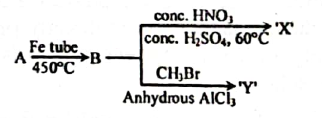
5. (i)
(ii) Monochloro alkane containing 2-Carbon
B, C and D are organic compounds.
Barishal Board 2017
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো