Chemistry 2nd Paper CQ-Mymensingh Board-2023
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
2. 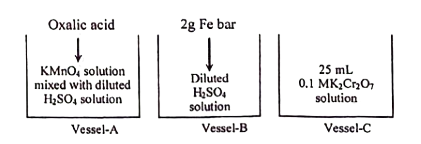 [The solution of vessel ' ' can be oxidized completely with the solution of vessel ' ']
[The solution of vessel ' ' can be oxidized completely with the solution of vessel ' ']
Mymensingh Board-2023
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
2.  [The solution of vessel ' ' can be oxidized completely with the solution of vessel ' ']
[The solution of vessel ' ' can be oxidized completely with the solution of vessel ' ']