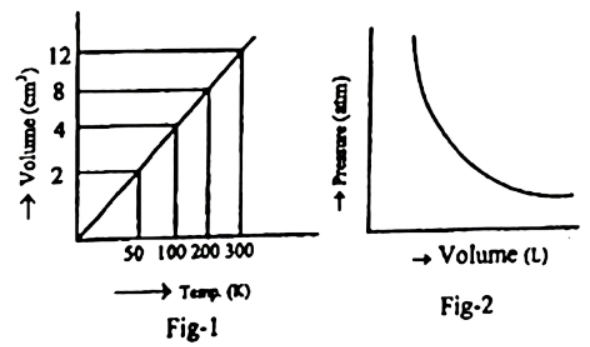
Chemistry 2nd Paper CQ-Dhaka Board-2021
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
1. 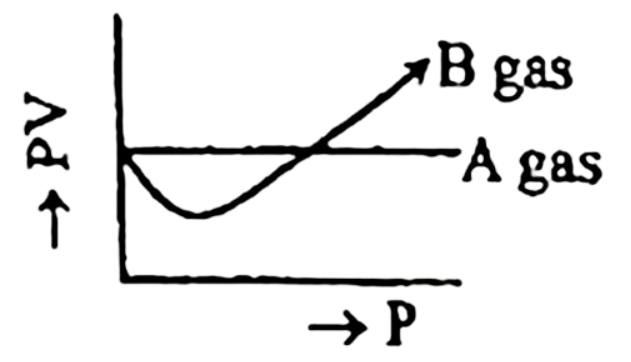 B gas responsible to consist green house gas.
B gas responsible to consist green house gas.
DB-21
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
3. 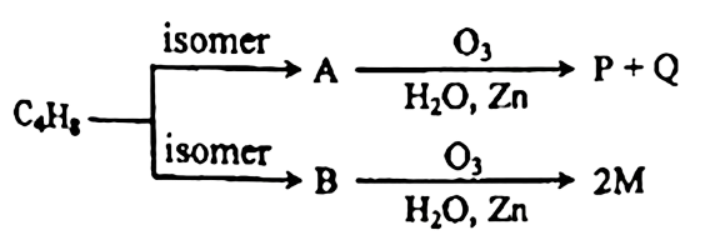 doesn't react with Tollen's reagent but reacts with that.
doesn't react with Tollen's reagent but reacts with that.
DB-21
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো