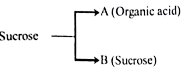Chemistry 1st Paper CQ-Dinajpur Board-2017
প্রশ্ন ৮·সময় ২ ঘণ্টা ৩৫ মিনিট
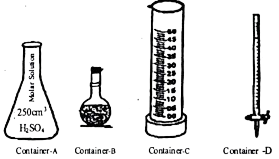
3. The atomic numbers of and elements are 6,7 and 8 respectively.
Din.B-17
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো
5. 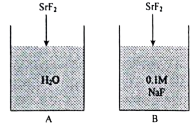 The solubility product of in container is
The solubility product of in container is
Din.B-17
ব্যাখ্যা আনলক করতে চর্চা প্রিমিয়াম এ আপগ্রেড করো