Chemistry 1st Paper MCQ-Jessore Board-2023
1.
For the above reaction-
i. if temperature is increased, the equilibrium will shift to the right
ii. increase of pressure has no effect on equilibrium
iii. increase of concentration of reactants may increase the backward rate of reaction
Which one is correct?
2. 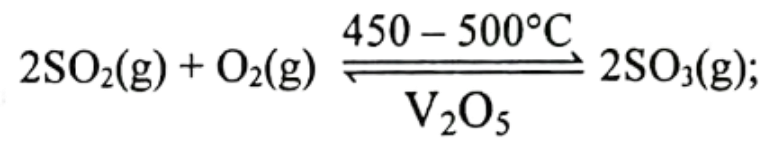 Characteristics of the reaction are-
Characteristics of the reaction are-
i. volume contraction occure in forward reaction
ii. excess amount of O₂ addition may change the equilibrium towards left direction
iii. backward reaction would be endothermic
Which one is correct?
3. Which one of the following is anti-oxidant agent?
4. Which one of the following is the correct order of the radius-
Period | 1 | 2 | 3 | 4 | 5 |
Elements of group 2 | - | X | Y | Z | Ba |
For element 'Z'-
i. oxidation number is changeable
ii. the value of magnetic quantum number of two electrons in the last shell be different
iii. Ammonium oxalate is used for the identification of ion
Which one is correct?

